🇫🇷 Lire en Français | 🇪🇸Leer en Español | 🇧🇷Leia em português
You have clear DNA results on both potential parents. So it is safe to breed them, right? Not necessarily. A health panel tells you what two animals are not. A genetic pairing matrix tells you what they can produce together.
Genomic data opens a door pedigree alone cannot. You can measure the actual genetic relationship between two animals, predict their offspring’s inbreeding coefficient, and see which carrier alleles overlap. This is the shift from testing individuals to planning litters.
A genetic pairing decision matrix produces one recommendation: breed, breed with caution, or do not breed.
- TL;DR
- Why DNA panels alone do not guarantee safe pairings
- Genomic COI reveals what pedigree COI hides
- Hitchhiking alleles: selecting for one trait can drag problems along
- Building your genetic pairing decision matrix step by step
- Tools and records for systematic pairing decisions
- When the matrix says stop and what to do next
- When to bring your veterinary geneticist into the conversation
- Conclusion
TL;DR
- Health panels identify carrier status but do not account for inbreeding or hitchhiking alleles; a genetic pairing matrix bridges this gap.
- Genomic COI reveals hidden kinship that pedigree COI misses, especially in small populations.
- A five-step matrix integrates genomic COI, carrier overlap, offspring COI projection, phenotype, and breed constraints into one pairing score.
- Both dogs and cats use the same framework; carrier frequencies and breed-specific haplotypes differ.
- Red-flag pairings are high COI plus overlapping recessive carriers. Consult a veterinary geneticist if you cannot resolve both risks.
- A pairing worksheet documents your decision and protects your program records.
- The genetic pairing decision matrix turns raw data into a teachable, repeatable process.
Why DNA panels alone do not guarantee safe pairings
The all-clear trap: when health panels miss the bigger picture
A health panel tests each animal individually. It answers: does this dog or cat carry mutation X? But a breeding decision is about what two animals produce together. Two “clear” individuals can still produce high-COI offspring if they share too many ancestors. High COI increases the probability that hidden recessive mutations will emerge homozygous in offspring.
A pairing that passes the panel test but fails the COI test introduces genetic drift and slow erosion of the gene pool. Your goal is to preserve your breed’s future health.
Your veterinarian can help interpret what “clear” means in context. A clear result from a small population carries less reassurance than the same result in a diverse breed.
| Decision Method | Assesses Individual Health | Assesses Offspring Risk | Detects Hidden Kinship |
|---|---|---|---|
| Health panel only | Yes | Partial (carriers only) | No |
| Pedigree COI only | No | Partial (assumes known pedigree) | No |
| Genomic COI only | No | Partial (inbreeding only) | Yes |
| Full decision matrix | Yes (via panel) | Yes (COI + carrier overlap) | Yes (genomic COI) |
Genomic COI reveals what pedigree COI hides
Pedigree COI is calculated from documented ancestry. It is simple and has been the gold standard for over a century. But it has three blind spots: incomplete records (especially in cats), conservative estimates (missing genetic material from undocumented ancestors), and inability to catch cryptic kinship where animals carry identical DNA blocks from remote common ancestors.
Genomic COI compares actual DNA. It identifies the percentage of the genome that is identical by descent, regardless of pedigree documentation. Two dogs with identical genomic COI can have very different pedigree COI if records differ. Genomic COI is the truth; pedigree COI is the best guess documentation allows.
Genomic testing is especially valuable in cat breeding, where pedigree databases are less comprehensive than in dogs. Your veterinarian can recommend testing when records are unclear or kinship needs verification before a pairing.
| Measure | Pedigree COI | Genomic COI |
|---|---|---|
| Based on | Documented ancestry | Actual DNA similarity |
| Detects cryptic kinship | No | Yes |
| Requires complete records | Yes | No |
| Can be wrong if | Records are incomplete or falsified | Rarely; DNA is the source of truth |
| Best use in dog breeding | Supplement to genomic testing | Primary measure |
| Best use in cat breeding | Helpful if available | Essential if records are incomplete |
Hitchhiking alleles: selecting for one trait can drag problems along
When you breed for a specific phenotype, you select for genes that produce that appearance. But sometimes, the alleles you want are physically close to alleles you do not want, a phenomenon called linkage. Selecting for your target trait can unintentionally concentrate a nearby hidden recessive mutations. For example, selecting for coat color may inadvertently enrich for a recessive eye disease on the same chromosome. The mutation comes along for the ride. Selecting for extreme phenotypes (very large, very small, unusual coats) narrows faster than numbers alone predict.
The matrix approach handles this by tracking haplotype diversity, not just individual alleles.
| Warning Sign | Normal Diversity | Narrowing Diversity |
|---|---|---|
| Carrier frequency in population | Varies by allele | Rising for same alleles |
| Phenotype variation in litters | Wide range | Very uniform |
| Unrelated parents have overlapping carriers | Rare | Frequent |
| Selected trait appears in >90% of program | No | Yes |
| New recessive disorders emerging | Expected background rate | Rising with each generation |
Building your genetic pairing decision matrix step by step
Step 1 and 2: Genomic COI and carrier status mapping
Gather two pieces of data: the genomic COI of each parent and their carrier status across relevant recessive disorders. A Labrador Retriever from a diverse population might score 8 to 12 percent; a dog from a small population might score 18 to 25 percent. For cats, expect 15 to 30 percent.
Carrier status requires a breed-specific health panel tests. Create a simple matrix: list each recessive allele, and mark whether each parent is clear, a carrier, or affected. Where both parents are carriers for the same allele, you have carrier overlap risk. Your veterinarian helps identify which overlaps are highest priority.
| Data Needed | Where to Get It | How Often to Update |
|---|---|---|
| Genomic COI for sire | Veterinary lab or direct-to-consumer genetic test | Once per animal; retest only if disputed |
| Genomic COI for dam | Veterinary lab or direct-to-consumer genetic test | Once per animal; retest only if disputed |
| Health panel for sire | Breed-specific genetic screening | At least once before first breeding |
| Health panel for dam | Breed-specific genetic screening | At least once before first breeding |
| Breed-specific carrier frequencies | Genetic research literature or breed database | Review annually; update if new data available |
| Pedigree records (both animals) | Stud book or breed registry | Maintain for all animals in program |
Step 3: Projecting offspring COI before you commit
Once you know each parent’s genomic COI, estimate offspring’s inbreeding coefficient. The formula is: projected offspring COI = (sire COI + dam COI) divided by 2, plus any additional COI from the pairing. If both parents have 10 percent COI and are unrelated, offspring will have approximately 10 percent COI.
High offspring COI increases the risk of recessive disorders appearing and reduces the population’s ability to respond to new health challenges. Most geneticists recommend keeping offspring COI below 5 percent to maintain genetic diversity. Beyond 10 to 12 percent, your program incurs genetic debt.
Your veterinarian interprets what offspring COI means for your breed and population size.
| Projected Offspring COI | Risk Level | Recommendation |
|---|---|---|
| Below 5% | Low | Proceed if carrier overlap is minimal |
| 5% to 7% | Moderate | Acceptable for isolated specific goals; monitor litter for recessives |
| 7% to 10% | Elevated | Consider an outcross alternative; if breeding, test all offspring genomically |
| 10% to 15% | High | Only if breed standard forces it and parents have zero carrier overlap |
| Above 15% | Very high | Do not breed. Invest in outcross or population restoration. |
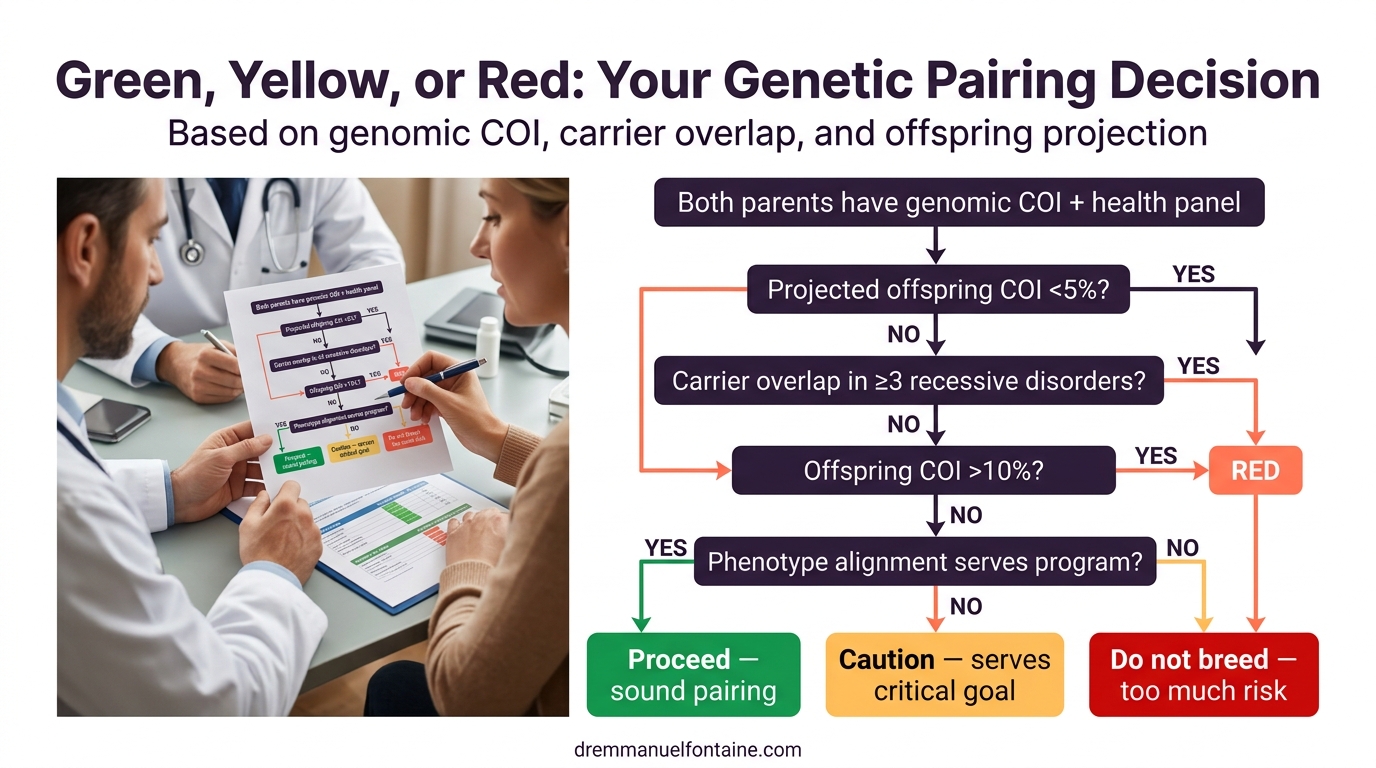
Steps 4 and 5: Phenotype scoring and the final pairing decision
A matrix is not just about numbers. Ask whether this pairing moves your program in the direction you intend. For each goal trait (movement, coat quality, temperament, function), rate each parent as “strong,” “average,” or “weak.” A strong dam with a weak sire might produce average offspring, acceptable if that trait is not your top priority.
Assign numerical weights to each factor. Genomic COI might be 40 percent, carrier overlap 30 percent, offspring COI 20 percent, and phenotype scoring 10 percent. Adjust to match your priorities.
The final decision is categorical: green (proceed; sound), yellow (caution; serves critical goal), or red (do not breed; too much risk). Your veterinarian helps contextualize numbers. High COI might be acceptable for a rare breed, but not in a breed with many alternatives.
| Factor | Green (Proceed) | Yellow (Caution) | Red (Stop) |
|---|---|---|---|
| Projected offspring COI | Below 5% | 5% to 10% | Above 10% |
| Carrier overlap (recessive) | None or rare | Moderate (1-2 overlaps) | Extensive (3+ overlaps) |
| Genomic COI of parents | Below 8% | 8% to 12% | Above 12% |
| Phenotype alignment | Both strong for goals | One strong, one average | Misaligned with program |
| Haplotype diversity | High diversity | Moderate diversity | Limited diversity |
Tools and records for systematic pairing decisions
Which genomic tests to request from your vet
You can test over 300 genetic variants in dogs and over 40 in cats. Start with your veterinarian and breed club for recommendations on which disorders matter most.
For dogs, multibreed panels test 30 to 100 recessive disorders; breed-specific panels test only the 10 to 20 most relevant. For cats, breed-specific panels are essential because different breeds have different concerns.
Genomic COI testing is offered separately or combined with health panels. Testing COI on all potential breeding candidates before first breeding is most efficient. Your veterinarian helps negotiate costs and ensures proper sample tracking.
| Test Type | Dogs: Best Uses | Cats: Best Uses |
|---|---|---|
| Multibreed health panel (30-100 variants) | new to a breed, research | Less common; most cat labs offer breed-specific |
| Breed-specific health panel | Establish carrier status in breeding program | Most useful; focus on breed-relevant disorders |
| Genomic COI test | Before first breeding; periodic population check | Essential if pedigree records incomplete |
| Parentage testing | Resolve disputed kinship; verify stud service | Useful in both species |
| Haplotype analysis | Research or advanced breeding programs | Research only; limited commercial availability |
Building a breed-specific carrier frequency reference
A frequency table tells you what percentage of your breed population carries each recessive allele. If 15 percent of your breed carry a recessive, then two unrelated animals have a 2.25 percent chance of both being carriers (15 percent times 15 percent). Overlapping carriers for a rare recessive are less concerning than overlapping carriers for a common one.
Carrier frequencies are the hidden denominator in pairing decisions. Build your reference table from breed club data, health foundation databases, and breed registries. Organize by allele, recording allele name, prevalence in carriers and affected animals, and data source. Update annually as new animals are tested.
Your veterinarian can help access published frequency data or organize breed club data. A carrier frequency table is a living document that improves with scale and time.
| Recessive Disorder | Dogs: Frequency in Breed (%) | Cats: Frequency in Breed (%) |
|---|---|---|
| Example: Collie Eye Anomaly (CEA) | 8-12% carriers in affected breeds | Not applicable (dogs only) |
| Example: Progressive Retinal Atrophy (PRA) | Varies 2-25% by breed | 4-8% in Siamese, Abyssinian |
| Example: Von Willebrand Disease Type 1 | 20%+ in Doberman, Pinscher | Not applicable (dogs only) |
| Example: Hereditary Sensory Neuropathy | 15-20% carriers in Pointer breeds | Not applicable (dogs only) |
| Example: Polycystic Kidney Disease | Not applicable (dogs only) | 5-10% carriers in Persians, British Shorthairs |
| Example: Hypertrophic Cardiomyopathy | Not applicable (dogs only) | Variable by breed; <1% to 5% |
The pairing decision worksheet your program needs
A pairing worksheet is the document you fill out every time you consider a breeding. It captures all data points from your matrix in one place and creates a permanent record of why you made the decision. A good worksheet includes sections for basic animal information (name, registration number, breed, age), genomic COI, health panel results, projected offspring COI, phenotype scoring, and final decision with rationale.
The worksheet forces you to move step-by-step through the matrix rather than skipping to a gut-feeling decision. It becomes evidence of your due diligence. Store worksheets in a secure location alongside pedigree records and genetic test results.
Your veterinarian may have templates or recommendations. Some breed clubs provide worksheets. Starting with a template and customizing it to your program’s priorities is faster than building from scratch.
| Worksheet Section | What to Record | Who Reviews It |
|---|---|---|
| Animal IDs | Name, registration #, breed, DOB | You, breed club on request |
| Genomic COI | Test result %, lab name, date | You, your vet, breed club |
| Health panel results | Carrier status for each disorder, test date | You, your vet |
| Projected offspring COI | Calculated %, formula used | You, your vet |
| Phenotype scores | Rating for each breed goal, rationale | You, mentors in your breed |
| Final decision | Green/Yellow/Red, full rationale, date signed | You, your vet, breed club records |
When the matrix says stop and what to do next
Red-zone pairings: high COI and overlapping carriers
A red-zone pairing has two or more of these characteristics: projected offspring COI above 10 percent, carrier overlap in three or more recessive disorders, or genomic COI above 12 percent in both parents. Red-zone pairings increase the odds of genetic problems substantially.
The most dangerous are pairings where both parents are carriers for a common autosomal recessive in your breed. Every offspring has a 25 percent chance of being affected. If you breed these animals, assume some offspring will be born with the disorder.
Not all red-zone pairings are equally problematic. High COI with zero carrier overlap might be defensible if it is the only way to preserve a critical lineage in a small population. But moderate COI with extensive carrier overlap is more concerning because you are stacking multiple genetic risks. When the matrix says red, ask why you are considering this pairing. If the answer is “no good alternative,” your breed has a population problem, not just a pairing problem.
| Pairing Profile | COI Risk | Carrier Overlap | Decision |
|---|---|---|---|
| Two unrelated animals, high genetic diversity | Low (below 5%) | Minimal or none | Green: Proceed |
| Unrelated, good diversity, 1-2 carrier overlaps for rare recessives | Low (below 5%) | Minimal | Green: Proceed |
| Half-siblings or cousins, no carrier overlap | Moderate (5-8%) | None | Yellow: Caution acceptable |
| Half-siblings or cousins, 2-3 carrier overlaps | Moderate (5-8%) | Moderate | Yellow or Red: Test all offspring |
| Close relatives (both parents COI >10%) with carrier overlap | High (>10%) | Extensive (3+) | Red: Do not breed |
Genetic bottleneck warning signs in your program
A genetic bottleneck happens when population genetic diversity shrinks dramatically, usually because too many animals descend from a small number of founders. Bottlenecks do not reverse quickly; restoring lost diversity takes many generations.
Early warning signs are subtle: carrier frequencies for recessives rising, unrelated animals increasingly showing carrier overlap, average COI in your program creeping upward, litter sizes shrinking, or fertility and vitality issues emerging. A bottleneck can be triggered by a single event (disease outbreak) or gradual pressure (selecting for extreme phenotypes, concentrating on successful bloodlines, or selective registration).
If you spot warning signs, act now, not later. Recovering lost diversity requires extreme measures: outcrosses, frozen sperm from retired animals, or crossing to a closely related breed. These are more disruptive than maintaining diversity from the start. Your veterinarian can help monitor trends over time, tracking average COI, carrier frequency, and litter size annually for early warning.
| Generation | Average COI in Program | Frequency: Common Carriers | Unrelated Pairs with Overlap |
|---|---|---|---|
| Generation 0 (founders) | 2-4% | Low (1-3%) | Rare |
| Generation 2-3 | 4-6% | Rising (2-5%) | Beginning to appear |
| Generation 5 | 6-10% | Elevated (3-8%) | Common; few alternatives |
| Generation 8+ | 10-15% | High (8%+) | Most pairings have overlap |
| Bottleneck recovery (requires outcross) | Returns to lower range | Decreasing post-outcross | Diversity restored |

When to bring your veterinary geneticist into the conversation
You do not need a specialist for every pairing decision. Many straightforward decisions (green pairing, unrelated animals, clear carrier results, aligned phenotypes) are manageable with your regular veterinarian and breed club mentors. But extensive carrier overlap, very small breed populations, conflicting COI results, or breed-wide genetic management programs require a board-certified veterinary geneticist.
Geneticists help navigate ethical dilemmas numbers alone cannot resolve. What if your breed is so small that all remaining animals are related? What if a critical bloodline carries a common recessive, and eliminating it eliminates the breed standard? A geneticist helps you think through trade-offs and long-term consequences.
Geneticists also design testing strategies for your breed, help prioritize which disorders to screen for, interpret carrier frequencies data, and develop breeding guidelines that balance genetic health with breed constraints. Ask your veterinarian or breed club for referrals.
| Situation | Your Vet Alone is Sufficient | Consultation with Geneticist Recommended |
|---|---|---|
| Single pairing decision with clear data and low risk | Yes | No |
| Program with >3 carrier overlaps or COI >10% | Limited; consult vet first | Yes |
| Breed population <200 animals | May lack context | Yes |
| Conflicting test results or parentage questions | Ask vet to help coordinate | Yes |
| Developing breed-wide genetic management plan | No | Yes |
| Annual routine pairing decisions in established program | Yes | No |
Want to put all of this into action before your next breeding decision? Inside the Breeder Vault, you will find the Genetic Pairing Decision Matrix Field Protocol: a printable scoring matrix with decision trees, carrier-frequency tables, and veterinarian consultation prompts, built so you can score any pairing in under 15 minutes. Take it into your next planning meeting and turn DNA data into confident, repeatable breeding decisions.
Conclusion
A genetic pairing decision matrix transforms raw DNA data into structured decisions. It replaces “we think they will work” with “here is why we chose this pairing, and here is what we are monitoring.” That shift builds a breeding program you can defend and improve over time.
The matrix combines genomic COI, carrier status mapping, offspring COI projection, phenotype complementarity, and breed constraints. Together, they paint a picture of genetic risk and breeding goal alignment that guides your decision.
For both dog and cat breeders, the process is the same. The framework is universal. Start with your veterinarian. Get genomic COI and health panel results. Map carrier overlap. Calculate projected offspring COI. Score phenotype alignment. Then ask whether this pairing serves your program and respects your population’s long-term future. If the answer is yes, documented in a worksheet, you have a defensible decision.
Genetic pairing decisions are part of a larger practice of stewardship: maintaining genetic diversity, tracking health outcomes, updating your carrier frequency reference, and being willing to step back when numbers say stop. That stewardship turns a genetic pairing decision matrix from an academic exercise into a tool that protects the animals you breed and the breeds you serve.


